Understanding the Common Ion Effect: A Comprehensive Guide
Welcome to this professional guide aimed at chemistry enthusiasts who seek to understand and master the concept of the Common Ion Effect. Whether you are a seasoned chemist, a student, or simply passionate about chemistry, this guide will provide you with step-by-step guidance, actionable advice, and practical solutions to unravel the intricacies of the Common Ion Effect. Our aim is to address your pain points, offer practical examples, and ensure you are equipped with the knowledge to apply this concept in real-world scenarios.
What is the Common Ion Effect?
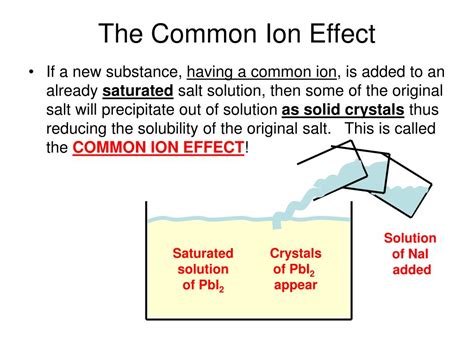
The Common Ion Effect occurs when a common ion is introduced to a solution, influencing the solubility equilibrium of a sparingly soluble salt. This effect highlights the relationship between solubility and ionization, which is pivotal in various fields like pharmaceuticals, environmental science, and industrial chemistry.
This phenomenon is often seen when salts containing a common ion are dissolved in a solution that already has that ion present. For example, adding sodium chloride (NaCl) to a solution containing a small amount of silver nitrate (AgNO3) will impact the solubility of the silver chloride (AgCl).
Problem-Solution Opening: Addressing User Needs
Many learners find the Common Ion Effect complex and challenging to grasp due to its nuanced interaction with solubility equilibrium. The challenge lies in understanding how the presence of a common ion can suppress the dissolution of a salt, thus affecting its solubility product. Our goal is to demystify this concept with practical examples, actionable advice, and problem-solving techniques that address these very challenges.
By breaking down the Common Ion Effect into understandable parts and offering real-world examples, this guide aims to equip you with the tools to not just understand but also apply this concept effectively.
Quick Reference
- Immediate action item: When adding a salt to a solution with a common ion, predict the shift in solubility using the solubility product constant (Ksp).
- Essential tip: To understand the effect, calculate the concentrations of the ions involved and determine the new equilibrium position.
- Common mistake to avoid: Overlooking the role of the common ion, which often leads to incorrect solubility predictions.
How to Predict the Common Ion Effect
To predict the Common Ion Effect, follow these detailed steps:
Step 1: Identify the common ion present in the solution. In our previous example, the common ion is chloride (Cl-) present in both sodium chloride (NaCl) and silver chloride (AgCl).
Step 2: Write the dissociation equations for all involved salts. For NaCl and AgCl, the dissociation would be:
- NaCl → Na+ + Cl-
- AgCl → Ag+ + Cl-
Step 3: Establish the equilibrium expression for the sparingly soluble salt. For AgCl, this is:
AgCl(s) ⇌ Ag+(aq) + Cl-(aq)
Ksp = [Ag+][Cl-]
Step 4: Calculate the concentrations of all ions at equilibrium. If the concentration of Cl- from NaCl is significant, it will affect the equilibrium.
Step 5: Apply the Common Ion Effect: Since the chloride ion is common, it increases the concentration of Cl- in the solution, shifting the equilibrium to the left (Le Chatelier’s Principle), thereby decreasing the solubility of AgCl.
Detailed How-To Sections
Calculation Example: Predicting Solubility with Common Ions
Let’s walk through a practical example to solidify your understanding:
Consider a solution containing 0.01 M NaCl and a small amount of AgNO3. We need to determine how the presence of Na+ and Cl- affects the solubility of AgCl.
Step 1: Write the dissociation equations:
- NaCl → Na+ + Cl- (0.01 M Cl- from NaCl)
- AgNO3 → Ag+ + NO3-
- AgCl → Ag+ + Cl-
Step 2: Determine the initial concentrations of all ions at equilibrium.
Since AgNO3 dissociates completely:
- Initial [Ag+] = x (assuming x moles of AgNO3
- Initial [Cl-] = 0.01 M (from NaCl)
Step 3: Apply the Ksp expression for AgCl:
AgCl(s) ⇌ Ag+(aq) + Cl-(aq)
Ksp = [Ag+][Cl-] = 1.8 × 10-10
At equilibrium:
- [Ag+] = x - s (where s is the amount of AgCl that dissolves)
- [Cl-] = 0.01 + s
Step 4: Since Ksp is small and s is very small (since AgCl is sparingly soluble), assume x ≈ s.
Ksp = (s)(0.01 + s) ≈ 1.8 × 10-10
Solving for s:
s ≈ √(1.8 × 10-10 × 0.01) ≈ 1.34 × 10-5 M
Thus, the solubility of AgCl in the presence of 0.01 M Cl- is drastically reduced to 1.34 × 10-5 M.
Advanced Considerations: Complex Salt Systems
When dealing with more complex systems, such as those involving multiple ions, the calculation becomes multi-step:
Step 1: Identify all common ions and write their dissociation equations. For instance, consider a mixture of KCl and CaCl2 where K+ and Cl- are common.
Step 2: Write the solubility equilibrium equations for each sparingly soluble salt. For CaCO3 and CaF2:
- CaCO3 → Ca2+ + CO32-
- CaF2 → Ca2+ + 2F
Step 3: Establish the Ksp expressions:
- CaCO3: Ksp = [Ca2+][CO32-]
- CaF2: Ksp = [Ca2+][F-]2
Step 4: Calculate the concentrations of the ions in the presence of common ions. For instance, if the initial [Cl-] from KCl is 0.02 M:
- [Ca2+] ≈ √(Ksp of CaCO3)
- [F-</