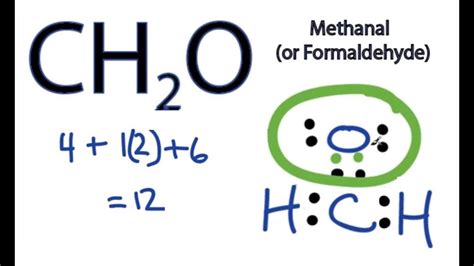
In the realm of chemistry, understanding molecular structures like the CH2O Lewis structure is crucial for grasping the foundational principles of molecular chemistry. This guide will break down the complexity of this seemingly simple molecule into digestible, practical information. This should prove invaluable for students, educators, and anyone fascinated by the science behind molecules.
The CH2O Lewis structure, representing formaldehyde, is a classic example that illustrates many core concepts of chemical bonding and molecular geometry. For anyone struggling with this topic, this guide is designed to provide step-by-step insights into the Lewis structure, offering practical advice and examples to enhance your understanding. Whether you’re a beginner or someone looking to refresh your memory, this guide will serve as a comprehensive resource.
Introduction: Why Understanding CH2O Lewis Structure Matters
The CH2O molecule, or formaldehyde, is fundamental in organic chemistry and chemical education. Understanding its Lewis structure provides insight into the molecule’s bonding, geometry, and reactivity. This knowledge is not just academic but also applicable in various fields, such as material science and environmental chemistry. By mastering the Lewis structure, you can better understand how atoms interact, how molecules form, and how chemical reactions occur.
Common Challenges
Many students find the CH2O Lewis structure daunting due to its seemingly simple yet intricate requirements. Common challenges include:
- Determining the correct number of valence electrons
- Distributing electrons to form the correct bonds
- Ensuring that all atoms achieve a stable octet
To tackle these challenges effectively, it’s essential to follow a structured approach and understand the underlying principles of electron sharing and molecular geometry.
Quick Reference Guide: Key Points for CH2O Lewis Structure
Quick Reference
- Immediate action item with clear benefit: Start with a rough sketch of the molecule. Draw carbon © in the center, surrounded by two hydrogen (H) atoms and one oxygen (O) atom. This visualization helps organize your approach.
- Essential tip with step-by-step guidance: Begin by totaling the valence electrons. Carbon has 4, hydrogen has 1 each (2 total), and oxygen has 6. This gives you 12 valence electrons to distribute.
- Common mistake to avoid with solution: Failing to place lone pairs on the oxygen atom can lead to an incorrect structure. Always remember that oxygen needs two lone pairs to satisfy its octet, and this will also allow for the correct double bond with carbon.
Detailed Steps for Drawing the CH2O Lewis Structure
To understand and draw the CH2O Lewis structure, follow these detailed steps:
Step 1: Determine Total Valence Electrons
Start by adding up all the valence electrons from each atom in the molecule. For CH2O:
- Carbon © has 4 valence electrons
- Each hydrogen (H) has 1 valence electron, so two hydrogens provide 2 valence electrons
- Oxygen (O) has 6 valence electrons
Adding these together, you get:
- Total valence electrons = 4 © + 2 (H) + 6 (O) = 12 valence electrons
Step 2: Sketch the Basic Structure
Visualize the molecule by placing the atoms in their typical bonding positions:
Arrange carbon in the center, with hydrogens on either side and oxygen adjacent to carbon:
O
|
H - C - H
This simple sketch helps in planning where electrons will be placed.
Step 3: Distribute Electrons and Form Bonds
Now distribute the 12 valence electrons to form bonds and lone pairs:
- First, form single bonds between carbon and each hydrogen:
- This uses up 4 electrons (2 per bond), leaving you with 8 electrons.
- Next, form a double bond between carbon and oxygen:
- This uses up another 4 electrons, leaving you with 4 electrons.
- Distribute the remaining 4 electrons as two lone pairs on the oxygen atom:
- Each lone pair takes up 2 electrons.
Your structure should now look like this:
O :||| H - C - H
The oxygen now has two lone pairs, and all atoms fulfill the octet rule.
Step 4: Check the Octet Rule
Ensure that each atom adheres to the octet rule:
- Hydrogen atoms are satisfied with their duet rule (two electrons).
- Carbon has 8 electrons around it (4 from bonds and 4 from lone pairs on oxygen).
- Oxygen has 8 electrons around it (4 from the double bond and 4 from lone pairs).
This confirms that the CH2O Lewis structure correctly satisfies the octet rule for each atom.
Practical FAQ: Common Questions and Answers
What if my initial structure doesn’t seem to work out?
If your initial structure doesn’t seem to work, recheck the total valence electrons and revisit the distribution. Often, starting from the skeleton structure with bonds first helps identify where adjustments are needed. Remember to consider the formal charges on each atom to find the most stable structure. This may involve moving electrons to form different types of bonds or placing lone pairs in different locations.
Why is the double bond between carbon and oxygen?
The double bond between carbon and oxygen forms because carbon and oxygen both need extra electrons to complete their octets. Carbon, with only four valence electrons, requires additional electrons to achieve a full outer shell, and oxygen, with six valence electrons, also needs two more electrons. By sharing four electrons (two from each atom), they form a double bond, fulfilling their octet rule.
Advanced Tips and Best Practices
Here are some advanced tips to refine your understanding and skills in drawing Lewis structures:
- Use Formal Charge Calculation: To determine the most stable structure, calculate the formal charges for each atom and see which arrangement gives the lowest overall formal charge. This helps ensure that your structure is as stable as possible.
- Practice with Isomers: Understanding different structural isomers (molecules with the same formula but different structures) helps deepen your understanding of bonding possibilities and molecular geometry.
- Explore Hybridization: Understanding the hybridization states of atoms (like sp, sp2, and sp3) can give you insights into the geometry and bond angles in molecules. For formaldehyde, carbon is sp2 hybridized, influencing its trigonal planar shape.
By integrating these advanced techniques into your study routine, you will gain a more nuanced understanding of molecular structures and their implications in chemistry.
Conclusion
Mastering the CH2O Lewis structure is a stepping stone to broader chemical knowledge. With practice, the rules governing electron distribution and molecular geometry will become more intuitive. Remember, the key is to take it step by step and not get discouraged by the initial complexity. Use this guide as a reference to continually refine your skills and deepen your understanding of molecular chemistry. Happy learning!