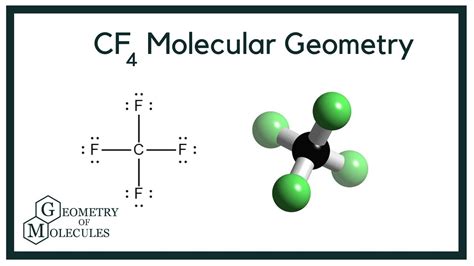
Understanding CF4 (Carbon Tetrafluoride) Molecular Geometry: A Comprehensive Guide
If you’ve ever felt lost while trying to grasp the intricacies of molecular geometry, especially with complex molecules like CF4, you’re not alone. Many students and professionals in the field of chemistry struggle to get their heads around the spatial arrangement of atoms in a molecule. Fear not! This guide is here to break down the molecular geometry of CF4 into digestible and actionable pieces of information.
We'll start with a step-by-step approach, incorporating real-world examples, to help you navigate through the complexities of CF4’s molecular structure. By the end of this guide, you’ll not only understand the geometry of CF4 but also be equipped with tips and best practices to tackle similar molecular geometries in your studies.
Immediate Action: Visualizing CF4 Geometry
Before diving deep into the details, let's take a moment to get a clear visual understanding. CF4 is a molecule with one carbon atom bonded to four fluorine atoms. The carbon atom is at the center, and the four fluorines are positioned symmetrically around it, akin to the vertices of a tetrahedron. This configuration is due to the carbon atom's sp3 hybridization, which ensures an even distribution of electron density around the central atom, minimizing electron repulsion.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Draw the Lewis structure of CF4 to visualize the distribution of atoms and bonds.
- Essential tip with step-by-step guidance: To determine the geometry, start with the VSEPR model. Predict the shape by calculating the total valence electron pairs and accounting for lone pairs.
- Common mistake to avoid with solution: Confusing the concept of hybridization with the molecular shape. Remember, hybridization (sp3 for CF4) determines the geometry, not the number of atoms directly.
Detailed Exploration: Understanding CF4 Molecular Geometry
Let’s delve deeper into the molecular geometry of CF4. This section will break down the process from determining the central atom to understanding the steric number and concluding the molecular shape.
Step-by-Step Guide to CF4 Geometry
Molecular geometry is all about understanding the shape of a molecule based on the arrangement of atoms around a central atom. For CF4, follow these detailed steps:
1. Identify the Central Atom
In CF4, carbon (C) is the central atom. Carbon is bonded to four fluorine (F) atoms. The placement of the central atom sets the foundation for the molecule's shape.
2. Draw the Lewis Structure
Drawing the Lewis structure helps visualize how electrons are distributed. Carbon has 4 valence electrons, and each fluorine has 7. The total valence electrons are 32 (4 from carbon + 28 from four fluorines).
Here's how to do it:
- Place carbon in the center and connect each fluorine atom with a single bond.
- Distribute the remaining electrons to satisfy the octet rule for each atom. Carbon will share electrons with each fluorine to complete its octet.
Your Lewis structure should have carbon surrounded by four single bonds, each connecting to a fluorine atom.
3. Determine the Steric Number
The steric number tells you how many regions of electron density surround the central atom. For CF4, the steric number is 4 (four single bonds with fluorine atoms).
4. Apply the VSEPR Model
Using the Valence Shell Electron Pair Repulsion (VSEPR) model helps predict molecular geometry. According to VSEPR, electron pairs repel each other. In CF4, the four fluorine atoms arrange themselves as far apart as possible to minimize repulsion, creating a tetrahedral geometry.
5. Confirm the Geometry
With the VSEPR model, we can now confirm CF4’s molecular geometry. Since there are no lone pairs on the central atom, the molecular shape is the same as the electron pair geometry: tetrahedral.
Practical Application: How to Apply Your Knowledge
Understanding CF4's molecular geometry is just the first step. Here’s how to put this knowledge to use in practical scenarios:
To determine the geometry of similar molecules, follow these steps:
- Identify the central atom.
- Draw the Lewis structure.
- Calculate the steric number.
- Apply the VSEPR model to predict the shape.
- Determine the molecular geometry.
This method is universally applicable, not just to CF4, but to many other molecules you’ll encounter.
Common Misconceptions and Pitfalls
Even with a solid understanding, there are common pitfalls to watch out for:
- Misunderstanding the role of hybridization: Remember, hybridization defines the geometry by arranging electron pairs around the central atom. Don’t confuse it with counting the number of atoms directly.
- Neglecting lone pairs: In molecules with lone pairs, their presence significantly affects the molecular shape. Always account for them when applying the VSEPR model.
- Overcomplicating the VSEPR model: Keep it simple. Start by predicting based on the steric number and refine based on the actual electron distribution.
FAQs on CF4 Molecular Geometry
What is the bond angle in CF4?
The bond angle in a tetrahedral molecule like CF4 is approximately 109.5 degrees. This angle is characteristic of tetrahedral geometry, where four electron pairs (or bonds) are distributed evenly around the central carbon atom.
Why is CF4 nonpolar despite having polar C-F bonds?
CF4 is nonpolar due to its symmetrical tetrahedral shape. The dipole moments of the four polar C-F bonds cancel each other out because they are evenly distributed around the central carbon atom. This symmetry results in a net zero dipole moment, making the molecule nonpolar.
By understanding these fundamental principles and following the outlined steps, you’ll have no trouble mastering the geometry of CF4 and applying this knowledge to other molecules.