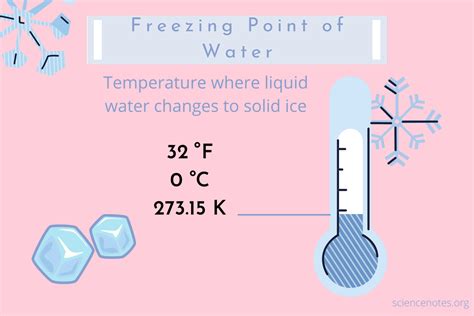
Understanding the freezing point of water at Celsius is crucial for scientists and students alike, especially when dealing with experiments that require precise temperature control. This guide aims to break down the concepts of freezing points, offer actionable advice, and provide practical examples to help you master this fundamental principle.
In science, understanding the freezing point is vital because it underpins various experiments, from chemical reactions to physical property studies. The freezing point of water, under standard atmospheric pressure (1 atm), is 0°C (32°F). This guide will walk you through essential steps to grasp this concept thoroughly, from basic principles to advanced applications.
Why Understanding the Freezing Point Matters
Grasping the freezing point of water is fundamental for many scientific disciplines. It helps in designing and interpreting experiments where temperature control is crucial. For instance, in biology, understanding how temperature affects enzyme activity or in chemistry, where precise temperature control can mean the difference between successful and failed synthesis reactions. Mastery of this concept can prevent costly mistakes and ensure the reliability of scientific results.
Quick Reference
Quick Reference
- Immediate action item: Calibrate your thermometer to ensure it accurately reads 0°C at the freezing point of water.
- Essential tip: Use a controlled environment like an ice-water bath to verify the freezing point and practice temperature regulation.
- Common mistake to avoid: Failing to account for impurities or atmospheric pressure variations that can alter the freezing point.
How to Calibrate Your Thermometer for Accurate Readings
Calibrating your thermometer is a fundamental step in ensuring accurate readings at the freezing point. Here’s a detailed step-by-step guide:
Step-by-Step Guide
1. Prepare an Ice-Water Bath:
Fill a large container with water and add a handful of ice cubes. Stir the mixture to ensure the ice and water are well-combined.
2. Insert Thermometer:
Place your thermometer in the ice-water mixture, ensuring that it is fully submerged but not touching the bottom of the container.
3. Wait for Stabilization:
Allow the thermometer to sit in the mixture for several minutes until it reads a stable temperature. Under normal atmospheric pressure, this should be around 0°C.
4. Calibration:
Compare the thermometer’s reading to the known freezing point of water (0°C). Adjust the calibration settings on your thermometer if it reads differently.
5. Verify Calibration:
To ensure accuracy, repeat the process multiple times and check for consistency. A reliable thermometer should consistently show 0°C in the ice-water bath.
Advanced Techniques for Temperature Regulation
When working with precise temperature controls beyond the basic freezing point, understanding advanced techniques can greatly enhance your experiments’ success. Here’s how to elevate your temperature control game:
Step-by-Step Guide
1. Use of a Water Bath:
For more consistent and stable temperatures, use a water bath. Fill a larger container with water and use a smaller container or beaker for your experiment. Ensure the water covers the beaker adequately.
2. Submerging Equipment:
Submerge thermometers, reaction vessels, and other equipment in the water bath. Adjust heating or cooling elements to maintain a stable temperature around the freezing point.
3. Monitor and Adjust:
Constantly monitor the temperature. Use precise heating/cooling devices to maintain it at 0°C or below, as required by your experiment.
4. Employ Insulating Materials:
To prevent heat exchange with the environment, use insulating materials such as foam or vermiculite around your setup. This minimizes fluctuations due to external conditions.
5. Precision Instruments:
Invest in precision instruments like temperature controllers or programmable freezers if you are working on large-scale or highly precise experiments.
Common Pitfalls and How to Avoid Them
Even with best practices, some common pitfalls can occur. Here’s how to sidestep them:
Step-by-Step Guide
1. Ignoring Atmospheric Pressure:
Remember that atmospheric pressure can affect the freezing point. Altitude changes can slightly elevate or depress this point. Always account for these changes in high-altitude experiments.
2. Not Stirring Mixtures:
When working in ice-water baths or other controlled environments, ensure thorough mixing to promote even temperature distribution. Uneven mixing can lead to localized overheating or undercooling.
3. Failing to Use Pure Water:
Impurities in water can lower its freezing point. For precise experiments, use distilled or deionized water to avoid these interferences.
4. Misreading Thermometers:
Ensure you are reading the thermometer correctly. Look at eye level to avoid parallax errors. Also, ensure it is fully submerged without touching any container’s base.
Practical FAQ
What if my thermometer does not read 0°C in an ice-water bath?
If your thermometer does not read 0°C in the ice-water bath, it might need calibration. Check the manufacturer’s instructions for proper calibration procedures. Additionally, verify that your thermometer is fully submerged and not touching the container’s bottom. If calibration does not resolve the issue, consider using a second, calibrated thermometer to confirm the readings.
How can I maintain a consistent freezing point for longer periods?
Maintaining a consistent freezing point over extended periods requires careful management of your setup. Use a water bath to provide a stable medium. Ensure that your setup is well-insulated to minimize external temperature influences. Regularly check and adjust heating or cooling devices to keep the temperature stable. For extended experiments, consider using a precision temperature controller to maintain exact conditions.
What are some alternatives to ice-water baths for precise temperature control?
For more precise and controlled environments, consider using a refrigeration unit or a temperature-controlled water bath. These systems provide a more consistent and stable temperature than simple ice-water baths, particularly useful in professional and advanced laboratory settings. Programmable freezers and chilling baths offer even greater control and can maintain exact temperatures without fluctuations.
By understanding the freezing point of water at Celsius and following these practical guidelines, you can enhance the precision of your scientific experiments and ensure reliable, reproducible results. This foundational knowledge is essential for both beginners and advanced practitioners in scientific research.