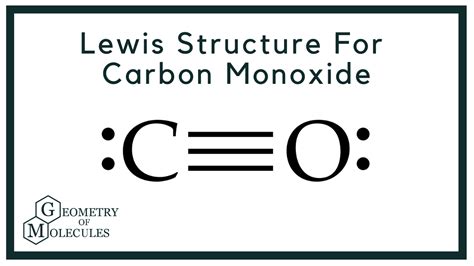
Understanding the Lewis structure of carbon monoxide (CO) is fundamental for comprehending its chemical properties and behavior in various reactions. Carbon monoxide is a colorless, odorless gas, with significant applications in both industrial processes and biological systems. Its Lewis structure provides critical insights into its bonding characteristics and reactivity.
Key insights box:
Key Insights
- Carbon monoxide’s Lewis structure reveals a triple bond between carbon and oxygen, highlighting its stability and reactivity.
- An understanding of formal charge and valence electrons is vital for drawing the accurate Lewis structure.
- Knowing the Lewis structure aids in predicting the molecule’s behavior in reactions and its role in various biological processes.
Delving into carbon monoxide’s Lewis structure begins with an examination of its constituent elements, carbon and oxygen. Carbon, with an atomic number of 6, possesses four valence electrons, while oxygen, with an atomic number of 8, has six valence electrons. In CO, carbon forms a triple bond with oxygen, a bond type characterized by the sharing of three pairs of electrons.
The construction of the Lewis structure for CO is methodical. Begin by placing carbon and oxygen atoms in a direct line, with carbon at one end. The total number of valence electrons available for the CO molecule is ten (four from carbon and six from oxygen). To achieve a stable octet for both atoms, three pairs of electrons are shared, resulting in a triple bond. The Lewis structure illustrates carbon on the left and oxygen on the right, each with two lone pairs on oxygen to fulfill its octet rule, leaving carbon without lone pairs.
This triple bond results in a linear geometry for the CO molecule. The formal charges must be considered in the Lewis structure. For CO, carbon and oxygen each achieve a formal charge of zero, ensuring the structure’s accuracy. The triple bond also implies sp hybridization on the carbon atom, which facilitates the molecule’s linear shape.
Analyzing the bonding in carbon monoxide involves understanding the nature of the triple bond. The triple bond consists of one sigma (σ) bond and two pi (π) bonds. The σ bond forms from the head-on overlap of an sp hybrid orbital from carbon and an sp hybrid orbital from oxygen. The two π bonds result from the side-by-side overlap of p orbitals from both carbon and oxygen.
The π bonds in CO are not equal in strength; one π bond is more stabilizing than the other due to the differential distribution of electron density. The stability of CO’s triple bond is crucial in understanding its reactivity, especially in reactions where the bond might break and form new bonds. The molecule’s triple bond makes CO a relatively stable yet reactive molecule, which is vital in both organic synthesis and biological systems, such as in hemoglobin binding.
In CO’s role within biological systems, its ability to bind with greater affinity to hemoglobin compared to oxygen underscores its toxicity. This binding results in the formation of carboxyhemoglobin, reducing the blood’s oxygen-carrying capacity and leading to carbon monoxide poisoning.
FAQ section:
Why is CO toxic?
CO is toxic because it binds more strongly to hemoglobin than oxygen, forming carboxyhemoglobin and reducing the blood’s oxygen-carrying capacity.
How does CO’s triple bond affect its reactivity?
CO’s triple bond is stable yet reactive, making it prone to break and form new bonds in various chemical reactions.
Understanding the Lewis structure of carbon monoxide allows for insights into its chemical properties and potential applications. Its triple bond not only illustrates its reactivity but also its pivotal role in both industrial and biological contexts. Mastery of CO’s Lewis structure is an essential step in comprehending its full chemical behavior.