Understanding the complex world of organic chemistry often involves grappling with terms that sound similar but have distinct meanings and roles. Two such terms are “amide” and “amine.” While they may seem interchangeable at first glance, they represent different classes of compounds with unique properties, reactions, and uses. This guide will help you grasp the fundamental differences between amides and amines, ensuring you can navigate this aspect of chemistry with confidence.
Introduction: Solving the Puzzle of Amides and Amines
Imagine you’re a chemist, and you’ve been given a molecular formula to decipher. You notice the presence of nitrogen atoms, and you’re unsure whether it signifies an amide or an amine. It’s a common conundrum that can lead to confusion and incorrect interpretations if not approached methodically. Amides and amines are both nitrogen-containing organic compounds, but their structures, reactivity, and applications differ substantially. This guide aims to demystify these differences, providing step-by-step guidance, practical solutions, and real-world examples that will arm you with the knowledge to distinguish between these two classes with ease.
Quick Reference
Quick Reference
- Immediate action item: Look at the functional group structure.
- Essential tip: Identify the presence of the carbonyl group in the compound.
- Common mistake to avoid: Confusing the amine's basicity with the amide’s neutral behavior.
Detailed How-To: Understanding Amides
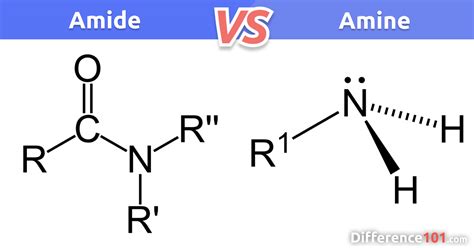
An amide is an organic compound that contains a carbonyl group (C=O) bonded to a nitrogen atom (N). The general formula for an amide is RCONR’2, where R and R’ can be hydrogen atoms or any other alkyl or aryl group. Amides are characterized by their relatively high stability and unique hydrogen bonding properties.
Here’s a step-by-step guide to help you understand amides:
- Identify the structure: An amide molecule has a carbonyl group (C=O) directly bonded to a nitrogen atom. This distinguishes it from amines, which do not have a carbonyl group attached to the nitrogen.
- Check for hydrogen bonding: Amides can form hydrogen bonds due to the presence of the nitrogen-hydrogen bond (if present). This property contributes to their stability and can be seen in the boiling points and solubility of amides.
- Understand reactivity: Amides are generally less reactive than amines. This reduced reactivity is due to the electron-withdrawing nature of the carbonyl group, which makes the nitrogen less available for nucleophilic attack.
- Explore applications: Amides have a wide range of practical uses. Polyamides, like nylon and Kevlar, are synthetic fibers renowned for their strength and durability. Other amides, like acetamide, are used as solvents and in pharmaceuticals.
Detailed How-To: Understanding Amines
An amine is an organic compound containing a nitrogen atom with a lone pair of electrons. Amines are derivatives of ammonia (NH3) where one or more hydrogen atoms are replaced by alkyl or aryl groups. The general formula for an amine is RNH2, where R can be hydrogen or an organic group.
To get a comprehensive understanding of amines, consider the following steps:
- Identify the basic structure: Amines have a nitrogen atom bonded to one or more alkyl or aryl groups. This basic structure allows them to act as nucleophiles and bases.
- Analyze basicity: Amines are generally basic compounds due to the presence of a lone pair of electrons on the nitrogen atom. The strength of the base depends on the electron-donating effects of the alkyl groups attached to nitrogen.
- Reactivity considerations: Amines are more reactive than amides due to the availability of the lone pair on nitrogen, making them good nucleophiles and enabling a variety of synthetic transformations.
- Real-world applications: Amines have numerous practical applications. They are used in the production of dyes, pharmaceuticals, and agricultural chemicals. Primary and secondary amines are crucial intermediates in the synthesis of many complex organic molecules.
Practical FAQ
What is the difference between primary, secondary, and tertiary amines?
In organic chemistry, amines are classified based on the number of alkyl or aryl groups attached to the nitrogen atom:
- Primary amine: One alkyl or aryl group is attached to the nitrogen atom (RNH2).
- Secondary amine: Two alkyl or aryl groups are attached to the nitrogen atom (R2NH).
- Tertiary amine: Three alkyl or aryl groups are attached to the nitrogen atom (R3N).
This classification affects the amine's properties, such as basicity and reactivity. For example, tertiary amines are generally less basic than primary and secondary amines due to steric hindrance reducing the availability of the lone pair on nitrogen.
How do you convert amides to amines?
Converting amides to amines typically involves a reduction reaction. One common method uses lithium aluminum hydride (LiAlH4) in anhydrous ether, which reduces the amide to an amine:
- Set up the reaction: Mix the amide with anhydrous lithium aluminum hydride in anhydrous ether.
- Heat gently to facilitate the reduction reaction.
- After the reaction is complete, carefully quench the reaction with water or an acidic solution to neutralize any remaining LiAlH4.
- Isolate and purify the primary amine product using standard workup techniques.
This reaction is highly effective for transforming amides into amines but requires careful handling due to the reactivity of LiAlH4.
Conclusion: Demystifying Amides and Amines
Mastering the differences between amides and amines is crucial for anyone delving into organic chemistry. By understanding their structures, properties, and practical applications, you can navigate the nuances of these compounds with ease. This guide provides a step-by-step approach to distinguish between amides and amines, with real-world examples and practical tips to enhance your comprehension. Whether you’re a student or a professional chemist, the insights here will serve as a reliable resource for tackling this essential aspect of organic chemistry.